Faculty
Lindsey "Les" Shaw
Professor & Associate Chair
Richard & Patricia Wood Endowed Chair in Natural Sciences
Director, Center for Antimicrobial Resistance
CONTACT
Office: ISA 6204
Phone: 813-974-2087
Lab: ISA 6014
Email
EDUCATION
- B.S. (hons) Microbiology, University of East Anglia (England)
- Ph.D. Molecular Microbiology, University of Sheffield (England)
- Postdoctoral Fellow, University of Georgia
- Research Assistant Professor, University of Missouri-Columbia
RESEARCH
- The Regulation of Virulence in Methicillin Resistant Staphylococcus aureus (MRSA)
- Molecular Epidemiology of S. aureus Infections
- Antibacterial Drug Discovery Targeting the ESKAPE Pathogens
Current Research
The Regulation of Virulence in Methicillin Resistant Staphylococcus aureus (MRSA): Staphylococcus aureus is a highly virulent and widely successful human pathogen that is among the most common cause of infectious disease and death in the United States. S. aureus is almost entirely unique amongst bacterial pathogens, as it can cause infection in almost every ecological niche of the human host. These infections range from the relatively benign, such as skin and soft tissue infections, boils, cellulitis and abscesses; to the systemic and life-threatening, such as endocarditis, septic arthritis, osteomyelitis, pneumonia and septicemia. The manner by which S. aureus achieves this is through the production of an arsenal of secreted and surface associated virulence factors. The production of each of these elements is tightly controlled at all tiers of the regulatory spectrum, allowing S. aureus to discretely titrate in or out the production of key virulence factors in a given niche. This then allows for the selective and disease specific regulation of pathogenic behaviors and provides a basis for control of the broad swath of infections caused by this organism. As such, a major focus of our work is to understand the dynamics of S. aureus infection, exploring how virulence factors are controlled within the host to specifically bring about the myriad disease states caused by this dangerous pathogen.
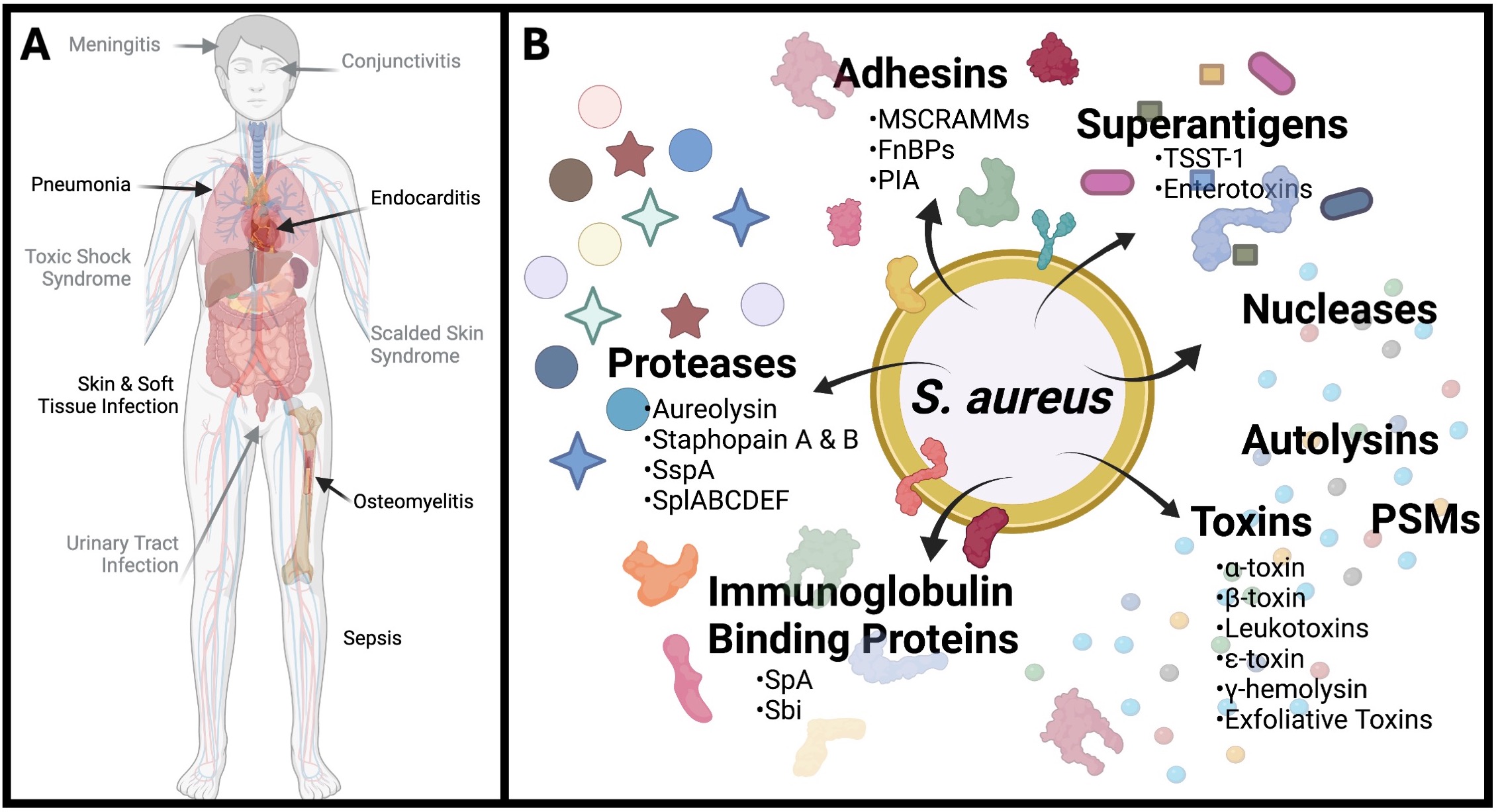
A: An example of the diverse infections S. aureus can cause in the human host. B: The myriad virulence factors S. aureus encodes to facilitate infection.
For example, transcription is controlled in bacterial cells using DNA binding proteins, two-component regulatory systems, and by alternative components of the RNA Polymerase complex. Our group has published studies on each of these processes, defining how they contribute to the manipulation of gene expression, and the progression of disease. The control of translation in bacteria is driven by regulatory RNA molecules. These are encoded within DNA like regular genes, but commonly do not specify a translatable product. Instead, they target other message bearing RNA molecules (mRNA) within the cell, and either stabilize or destabilize them. In this way, they lead to discrete, specific, and wide ranging effects on protein synthesis.
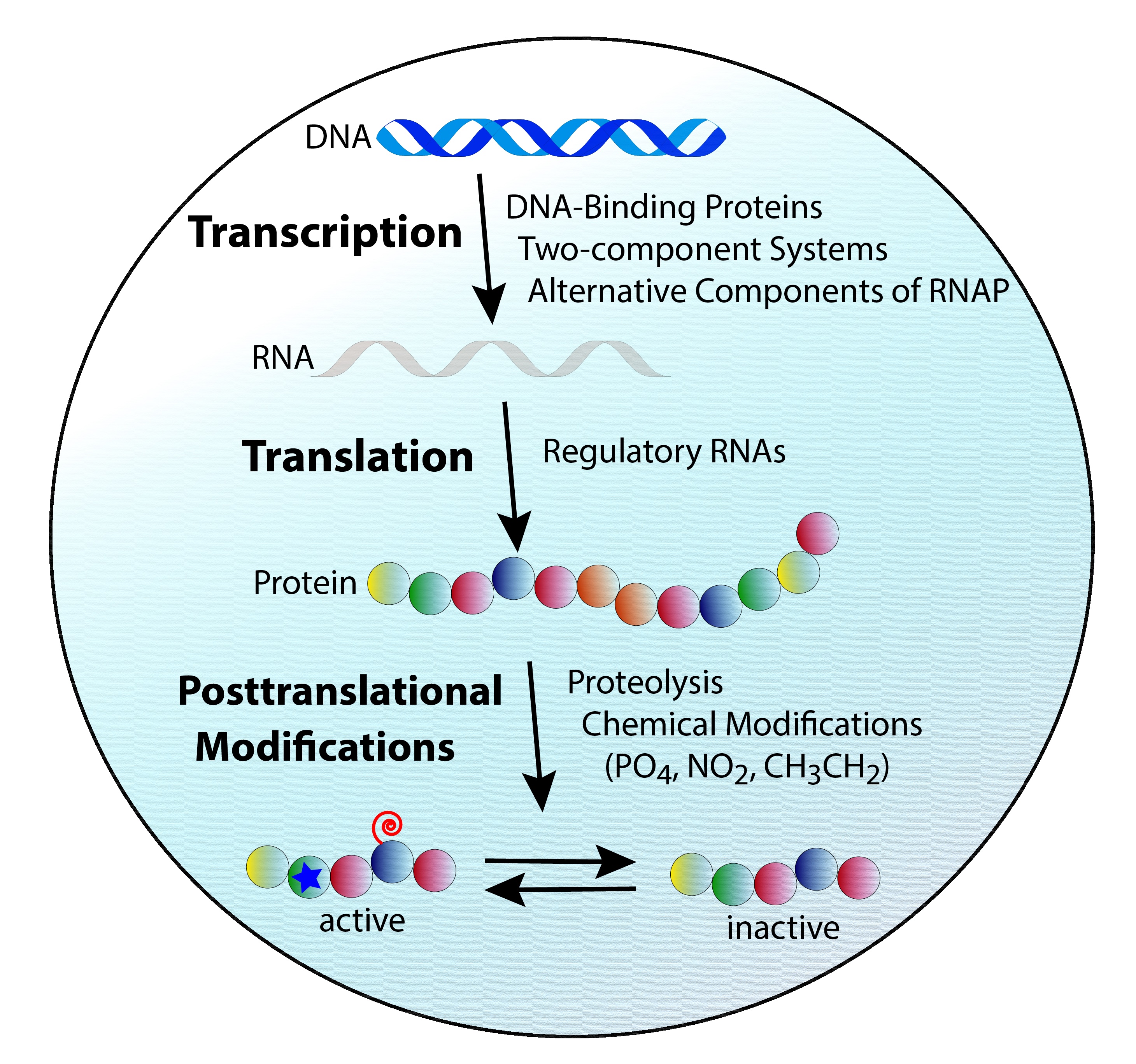
In the context of proteins, these are often made in an inactive form, requiring modification or processing to resulting in biological function. Conversely, some proteins are synthesized in a functional state, but must be specifically and discretely deactivated at key moments during growth. In the Shaw lab, we work on all kinds of these post-translation modifications, with a specific focus on how proteolysis, the cleavage of proteins by proteases, maintains cellular homeostasis, and facilitates the infectious process. Towards this latter point we have also developed cutting edge proteomic tools to study the impact of proteases on host-pathogen interaction. In so doing, we are shining important new light on exactly how bacteria use these vital enzymes to facilitate central processes such as nutrition, immune evasion, and movement to new sites of infection with the host.
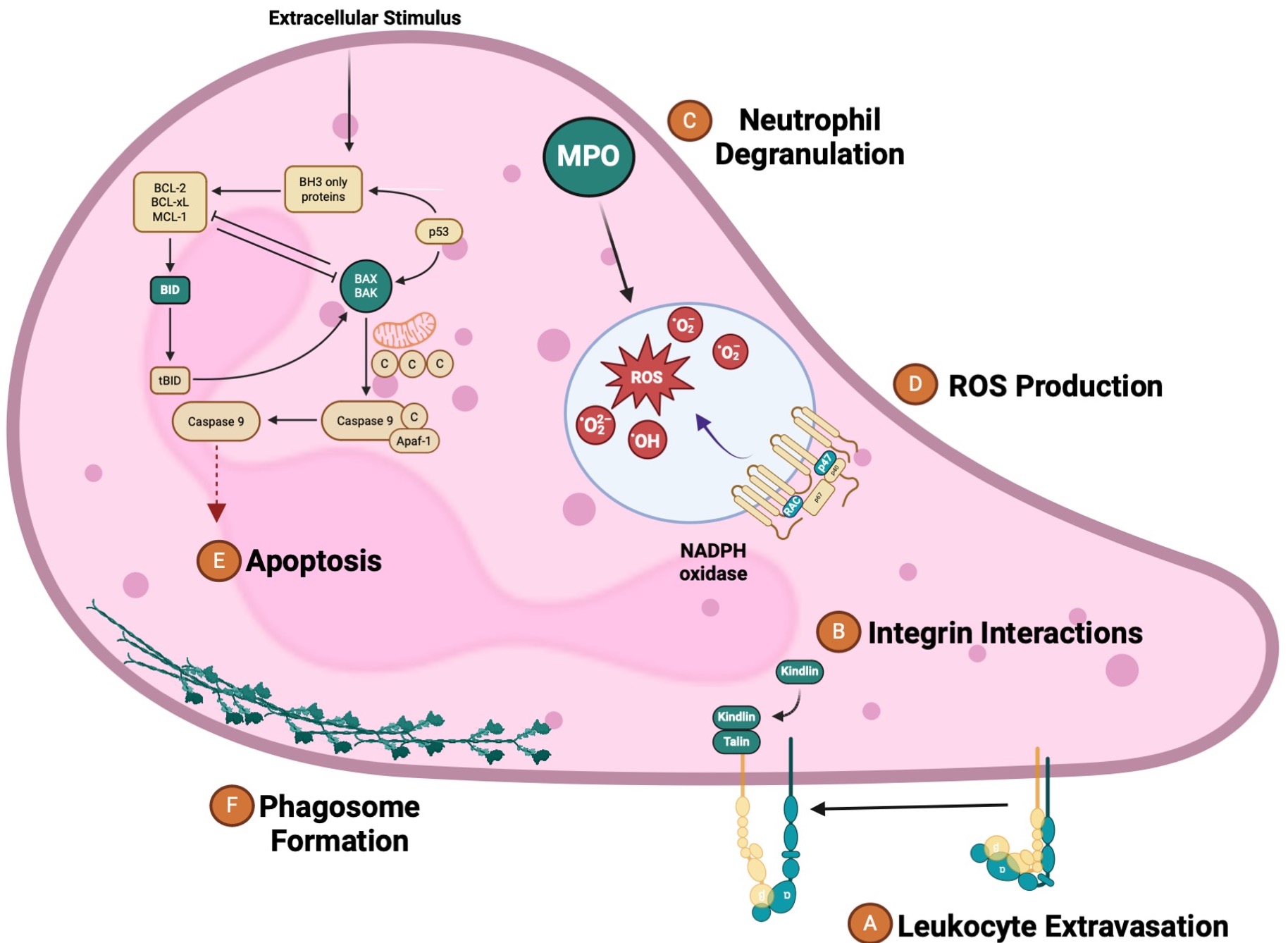
Figure: Engagement of S. aureus with Human Neutrophils
Molecular Epidemiology of S. aureus Infections: Another major arm of our work is studying natural variability in the pathogenic potential of S. aureus clinical isolates. Historically, S. aureus infections were confined to healthcare settings, afflicting the immunocompromised. However, over the last few decades, there has been a meteoric increase of severe invasive disease in healthy subjects lacking any predisposing factors. This trend shift is the result of, hypervirulent strains of MRSA that have evolved in the community (CA-MRSA). Of concern, these CA-MRSA strains have moved into clinical setting displacing existing hospital-associated MRSA isolates. As such, we work with infectious diseases clinicians in the USF College of Medicine and at Tampa General Hospital (TGH) to study the natural variability of circulating S. aureus isolates in the Tampa Bay region. Here, we collect strains from TGH, subject them to genome sequencing at the USF Genomics Institute, and characterize their infectious properties in the lab. We then correlate patient outcomes with our phenogenomic characterization of bacterial strains to gain new insight into the features that influence S. aureus infections in humans.
Antibacterial Drug Discovery Targeting the ESKAPE Pathogens: Despite the success of antimicrobial therapeutics in the past 70 years, infectious diseases remain the second-leading cause of mortality worldwide, causing 17 million deaths annually. Of this, the overwhelming majority are the result of bacterial pathogens. In the United States, there are almost 2 million hospital acquired infections each year, resulting in approximately 100,000 deaths. Perhaps the most significant public health concern in the context of bacterial infectious disease is the continued and rapid emergence of drug resistant strains during antibiotic treatment. Many bacteria are now unresponsive to conventional therapeutics, whilst still causing community and hospital acquired infections worldwide, leading to life-threatening and lethal diseases. Recently, the World Health Organization identified antimicrobial resistance as one of the three greatest threats facing mankind in the 21st century. As such, there is an undeniable and desperate need to develop new antibacterial therapeutics to fight the infections caused by these virtually untreatable pathogens. Unfortunately, the pace of drug resistance has outstripped the discovery of new antimicrobial agents, creating an urgent need for new antibiotics with novel mechanisms of action. The question is how we approach this, given that only 4 new classes of antibiotics have been marketed since 1970, and only 6 new antibiotics were approved by the FDA between 2003 and 2010. Indeed, there was a 75% decline in FDA approval for antibacterial agents from 1983-2007, largely the result of declining drug discovery efforts in industry. The is made even more concerning by the fact that only 4-5 companies are now seriously working on antimicrobial therapeutic development in the marketplace.
As such, in the Shaw lab, we work with a number of chemists and other research groups on the USF campus to identify new antimicrobial agents. These efforts are primarily focused on the ESKAPE pathogens (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumonia, Acinetobacter baumannii, Pseudomonas aeruginosa and Enterobacter cloacae). These are bacterial species that the CDC estimates cause more than two-thirds of all hospital-associated infections in the United States. They were identified by the Infectious Disease Society of America as causing the majority of infections in US hospitals, and having effectively managed to escape the activity of existing antimicrobial agents. Our work runs the entire spectrum of drug discovery, from hit identification, to lead development, in vivo efficacy testing, assessing anti-biofilm activity and mechanism of action studies.
GRADUATE STUDENTS
- Jessica Jackson
- Mary-Elizabeth Jobson
- Emily Felton
- Emilee Mustor
- Rachel Washburn
- Sara Daniels (MSPH student)
- Claudia Zamora Cisnero
- Julia Moncrieff
- Logan Desilets
- Allison Richards